
Introduction
Leave bare steel outdoors for a few weeks, and you'll watch it transform from clean metal to a rust-stained liability. That's structural failure in progress — not a cosmetic inconvenience.
For engineers and fabricators specifying components for industrial equipment, automotive assemblies, or infrastructure projects, unprotected metal means premature failure, costly downtime, and warranty claims.
Metal coatings are the engineered solution, but choosing the wrong coating for your application is an expensive mistake. Specify powder coating for a chemical immersion tank, and you'll see premature breakdown. Apply standard paint to offshore structural steel, and you're scheduling a maintenance repaint in five years instead of enjoying decades of protection.
This guide clarifies your options: what metal coatings are, the major types available, how they're applied, and how to match the right coating to your specific environment and performance requirements.
TLDR
- Metal coatings create protective barriers that prevent corrosion, extend service life, and cut maintenance costs
- Major types: powder coating (15–25 years), galvanizing (72+ years), electroplating, paint, anodizing, and thermal spray
- 75% of coating failures trace back to poor surface preparation, not coating chemistry
- Match coating to environment: galvanizing for wet/outdoor, powder coating for fabricated parts, anodizing for aluminum
- Correct coating choice cuts your share of the $2.5 trillion annual global cost of corrosion
What Are Metal Coatings?
Metal coatings are thin layers of material applied to a metal substrate's surface to serve protective, functional, or aesthetic purposes. They act as the metal's primary barrier against environmental damage, physical wear, and chemical attack. Without this barrier, oxygen, moisture, and corrosive chemicals reach the base metal directly, initiating oxidation and structural degradation.
Two Main Categories: Organic and Inorganic
Metal coatings divide into two fundamental categories based on their composition and bonding mechanism.
Organic coatings use polymer resins (epoxies, polyurethanes, acrylics, fluoropolymers) to form a barrier that sits on top of the metal surface, held in place through mechanical adhesion and chemical bonding. Liquid paints and powder coatings are the most common examples.
Inorganic coatings involve metallic deposition or electrochemical conversion of the substrate itself. Hot-dip galvanizing, electroplating, anodizing, and thermal spray all fall here. Rather than sitting on top, these coatings either metallurgically bond to the substrate or convert the surface layer into a protective oxide.
Beyond Rust Prevention: Functional Performance
Understanding these two categories makes it easier to see why coatings are specified far beyond basic rust prevention:
- Electroplated nickel improves wear resistance and reduces friction in moving parts
- Zinc-rich coatings on fasteners act as dry lubricants and torque control agents
- Anodized aluminum provides electrical insulation
- Specialized coatings satisfy regulatory requirements in food processing, medical devices, and aerospace where material certifications are mandatory
Metal Coatings vs. Metal Finishes: What's the Difference?
The terms "coating" and "finish" are often used interchangeably, but they describe different processes.
| Metal Coatings | Metal Finishes | |
|---|---|---|
| What it does | Adds a layer of a different material to the substrate | Alters the existing surface without adding new material |
| Examples | Powder coating, galvanizing, electroplating | Polishing, brushing, electropolishing |
| Result | New material layer (zinc, polymer resin, chromium) | Modified surface texture or smoothness |
Some processes blur this distinction. Anodizing is technically a finish because it converts the aluminum surface into aluminum oxide—but it's commonly called a coating because it creates a distinct protective layer. For practical purposes, if a process adds a new material layer, it's a coating. If it modifies the existing surface, it's a finish.
Common Types of Metal Coatings
Selecting the right coating requires understanding your substrate material, intended environment, performance requirements, and budget. Each coating type offers specific strengths and trade-offs. No single coating excels in every application.
Powder Coating
Powder coating involves electrostatically spraying dry thermosetting or thermoplastic powder onto a metal surface, then curing it in an oven to form a hard, smooth, durable layer. The electrostatic charge causes powder particles to adhere to the grounded metal part, creating uniform coverage even on complex geometries.
Key advantages:
- Wide variety of colors, textures, and gloss levels for aesthetic flexibility
- Virtually zero VOC emissions—no organic solvents required
- Up to 98% transfer efficiency with overspray reclamation, eliminating hazardous waste disposal costs
- 15 to 25 years of service life in exterior environments
- Cost-effective over time compared to liquid paint maintenance cycles
Powder coating is one of the most popular finishes for fabricated steel and aluminum components across industrial equipment, automotive assemblies, and architectural applications.
IMTS offers professional powder coating services as part of its end-to-end fabrication workflow, using a large 106"×60"×90" curing oven with complete in-house surface preparation—media blasting and iron phosphate chemical film washing—for enhanced coating adhesion.
Galvanizing
Galvanizing applies a zinc coating to steel through two primary methods:
Hot-dip galvanizing immerses fabricated steel into molten zinc (approximately 840°F), creating a metallurgically bonded coating specified under ASTM A123. The zinc layer acts as a sacrificial anode—it corrodes preferentially to protect the underlying steel, even if the coating is scratched or damaged.
Electrogalvanizing deposits zinc via electric current in a plating bath, producing thinner, more uniform coatings suitable for fasteners and sheet metal.
The American Galvanizers Association confirms that ¼-inch thick structural steel with standard zinc coating provides approximately 72 years of maintenance-free protection in harsh industrial environments. This "apply and forget" lifespan makes galvanizing ideal for structural steel, fencing, roofing, bridges, and outdoor infrastructure exposed to moisture where future maintenance is physically or economically prohibitive.
Electroplating
Electroplating uses electric current to deposit a thin metal layer—nickel, chromium, gold, copper, zinc—onto a conductive substrate. The part is immersed in an electrolyte bath containing dissolved metal ions. When current flows, metal ions migrate to the part's surface and form a uniform coating.
Benefits include:
- Improved wear resistance and reduced friction for moving parts
- Enhanced electrical conductivity for connectors and contacts
- Decorative appeal with bright, reflective finishes
- Corrosion protection through barrier and sacrificial mechanisms
Electroless plating offers a variant for complex geometries. Because it relies on chemical reduction rather than electric current, electroless nickel-phosphorus plating (ASTM B733) deposits with perfect uniformity across irregularly shaped parts, recesses, and internal passages.
It provides exceptional hardness (620–750 HK100) and is widely used in aerospace, automotive, and oil/gas applications.
Paint / Spray Coating
Liquid paint coatings—epoxy, polyurethane, acrylic, alkyd—are applied by spray gun, brush, or roller to create a protective and decorative barrier. Paint systems typically consist of a primer for adhesion and corrosion resistance, a midcoat for build and barrier properties, and a topcoat for UV resistance and aesthetics.
Considerations:
- Lower upfront cost compared to powder coating or metallic coatings
- Requires more frequent maintenance—typically 5 to 15 years between recoats in moderate to severe environments
- Surface preparation quality is absolutely critical to adhesion and longevity
- Solvent-borne paints emit VOCs and generate hazardous waste
Despite higher maintenance requirements, liquid paints remain essential for field touch-up and large structures that can't fit in powder coating ovens. High-performance epoxies also make them the go-to choice for specialized chemical resistance applications.
Anodizing
Anodizing is an electrochemical process primarily for aluminum that converts the surface into a hard, integrated aluminum oxide layer. The aluminum part is immersed in an acid electrolyte bath (typically sulfuric acid) and connected as the anode in an electrical circuit.
Oxygen ions released at the surface combine with aluminum atoms to form aluminum oxide directly within the metal itself.
Because the anodic oxide structure originates from the aluminum substrate itself, it is fully integrated with the underlying metal and physically cannot chip or peel like paint or plating. The porous oxide structure accepts dyes, allowing a wide range of colors while maintaining the metallic appearance.
MIL-A-8625 Types:
- Type I: Chromic acid anodizing for thin films with outstanding corrosion resistance in aerospace
- Type II: Sulfuric acid anodizing for general automotive, military, and electronics components
- Type III: Hardcoat anodizing for maximum abrasion and wear resistance in heavy machinery
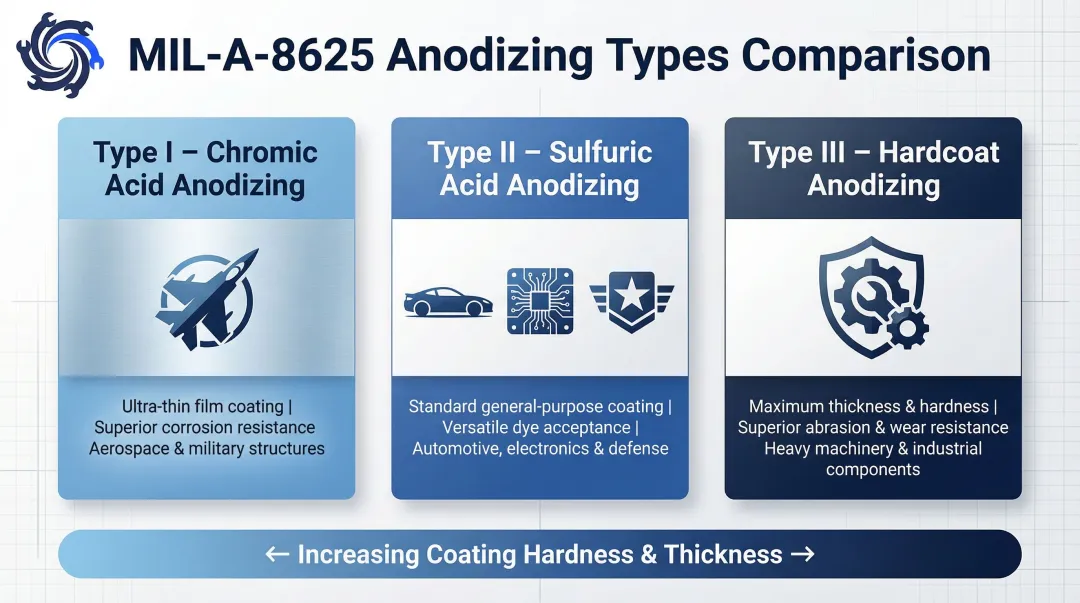
Anodizing is the standard finish for aerospace components, automotive trim, consumer electronics, and architectural aluminum where protection and aesthetics must coexist.
Thermal Spray Coating
Thermal spraying melts metallic or ceramic materials and propels them onto a substrate at high velocity. Methods include flame spray, wire-arc spray, plasma spray, and High-Velocity Oxyfuel (HVOF). The molten particles impact the surface, flatten, and rapidly solidify, building up a thick, mechanically bonded coating.
Applications:
- Rebuilding worn parts and restoring out-of-tolerance dimensions
- Applying heavy wear-resistant coatings to large components
- Replacing toxic hexavalent hard chrome plating with HVOF coatings
HVOF thermal spray achieves bond strengths exceeding 10,000 psi and provides superior density and wear resistance for high-value assets like shafts, bearing journals, cylinder bores, and heavy machinery components. The AWS C2.19/C2.19M standard governs thermal spray application for OEM manufacturing and repair.
Key Benefits of Applying Metal Coatings
Corrosion Resistance: Addressing the $2.5 Trillion Problem
Coatings create a physical barrier that prevents moisture, oxygen, and chemicals from reaching the base metal. Without this protection, metallic corrosion exacts a massive economic toll.
According to the NACE International (now AMPP) IMPACT study, the global cost of corrosion is estimated at $2.5 trillion annually, equivalent to 3.4% of global GDP. The study concludes that 15-35% of these costs—up to $875 billion globally—could be saved through currently available corrosion control practices. Protective coatings are the most direct and scalable way to capture those savings.
Extended Service Life and Reduced Maintenance Costs
A well-specified coating dramatically increases a component's lifespan, reducing replacement frequency and downtime. Consider the lifecycle comparison:
- Hot-dip galvanizing: 72+ years maintenance-free for structural steel
- Powder coating: 15-25 years for architectural applications
- Liquid paint: 5-15 years before maintenance recoat required
The long-term cost savings from extended service life often outweigh the upfront coating investment by a factor of 5 to 10. Procurement teams that shift from lowest-initial-cost purchasing to Total Cost of Ownership (TCO) models capture these savings and avoid premature asset failure.
Functional and Aesthetic Versatility
Modern coatings are engineered not just for protection but for specific performance needs:
- Electrical conductivity: Silver and gold plating for connectors
- Lubricity: PTFE and molybdenum disulfide coatings for low-friction applications
- Heat resistance: Ceramic and silicone coatings for exhaust systems
- Regulatory compliance: FDA-approved coatings for food processing equipment
Finish options—color, texture, gloss—support design and branding requirements across consumer and industrial products. Choosing the right system means balancing all of these needs upfront — getting the specification wrong can compromise both performance and compliance down the line.
The Metal Coating Process: Step by Step
Step 1 — Surface Preparation: The Foundation of Coating Success
Surface preparation is the most critical step in the coating process. Industry data confirms that 75% of all coating failures are caused by improper surface preparation—not coating chemistry.
All rust, grease, mill scale, and old coatings must be removed before application. Contaminants block adhesion, and without adequate surface profile—the peaks and valleys a coating needs to mechanically bond—even quality coatings fail prematurely.
Abrasive media blasting is the most effective prep method for achieving a clean, profiled surface. It simultaneously removes tightly adhered contaminants and creates the surface profile required for mechanical bonding—two goals accomplished in a single pass.
SSPC/NACE and ISO 8501-1 Cleanliness Grades:
- SSPC-SP 5 (White Metal) / Sa 3: 0% staining—free of all visible contaminants. Required for critical applications like chemical tanks and offshore platforms.
- SSPC-SP 10 (Near-White) / Sa 2.5: Maximum 5% random staining. Required for high-performance coatings in severe environments.
- SSPC-SP 6 (Commercial) / Sa 2: Maximum 33% random staining. Acceptable for standard structural steel in non-corrosive environments.

IMTS offers professional media blasting services using glass bead and coal slag to prepare metal components for coating. The integrated surface preparation workflow includes iron phosphate chemical film washing for enhanced coating adhesion and corrosion protection.
Step 2 — Coating Application: Precision Control for Uniform Coverage
Application method varies by coating type, and each requires precise control over temperature, thickness, and uniform coverage:
- Hot-dip galvanizing: Immersion in molten zinc at 840°F
- Powder coating: Electrostatic spray gun with grounded part
- Liquid paint: HVLP spray gun, airless spray, or brush
- Electroplating: Electric current in controlled electrolyte bath
- Thermal spray: High-velocity spray of molten particles
Coating thickness must meet specification requirements—too thin provides inadequate protection, too thick can cause brittleness, cracking, or dimensional tolerance issues.
Step 3 — Curing and Hardening: Locking in Performance
Most coatings require a curing phase to achieve full performance properties:
- Powder coating: Baked in oven at 350-400°F for 10-20 minutes
- Liquid coatings: Air-dry, heat-cure, or UV-cure depending on chemistry
- Electroplating: Post-treatment rinses and drying to remove residual chemicals
- Anodizing: Sealing in hot water or chemical bath to close pores
Once curing is complete, the part moves to inspection—the final checkpoint before delivery.
Quality Inspection: Verifying Specification Compliance
Reputable fabricators conduct post-coating inspection to verify that the coating meets specification before delivery or assembly:
- Adhesion testing: Cross-hatch or pull-off tests per ASTM standards
- Thickness measurement: Magnetic gauges for ferrous substrates, eddy current gauges for non-ferrous
- Visual inspection: Color, gloss, uniformity, and defect identification
Comprehensive inspection is a standard part of professional metalworking service. IMTS provides full inspection reports using advanced metrology equipment including portable and stationary CMM systems for dimensional verification before and after coating.
How to Choose the Right Metal Coating for Your Project
Match the Coating to the Environment
Environmental exposure is the primary factor determining coating selection. Mismatching coating to environment is the most common and costly specification mistake.
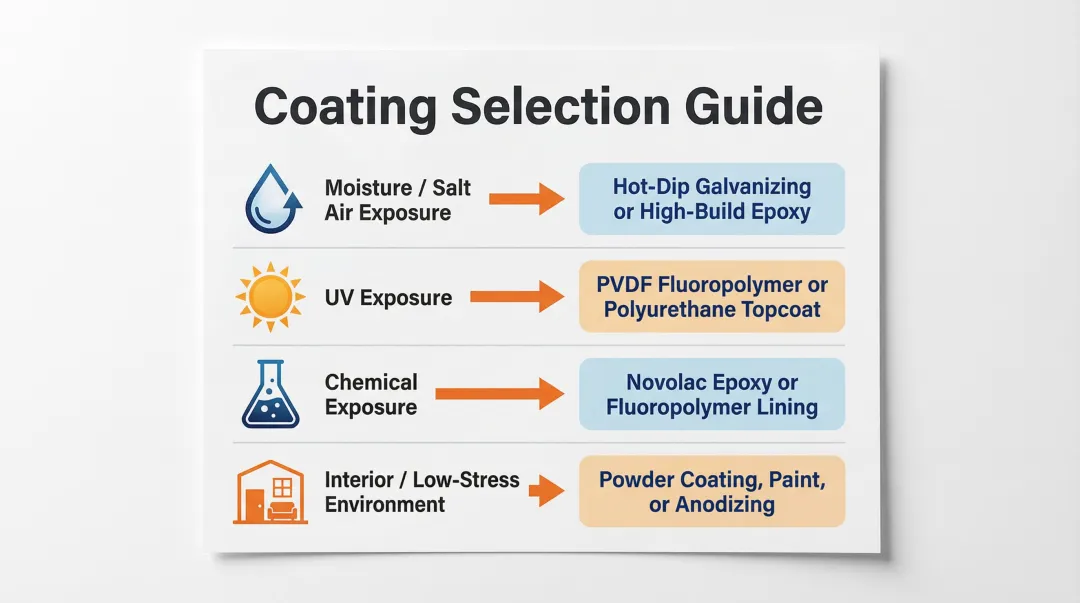
Key environmental factors to evaluate:
- Moisture/salt air exposure: Favor hot-dip galvanizing (ASTM A123) or high-build epoxy systems
- UV exposure: Favor PVDF fluoropolymer (AAMA 2605) or polyurethane topcoats with UV stabilizers
- Chemical exposure: Favor Novolac epoxy or fluoropolymer linings for immersion resistance
- Interior/low-stress applications: Standard powder coating, paint, or anodizing may suffice
The ISO 12944 standard categorizes environments from C1 (Very Low / heated interiors) to C5 (Very High Industrial) and CX (Extreme Offshore). Specify coating systems that meet or exceed the corrosivity category for your application.
Consider Substrate Compatibility
Not all coatings work on all metals. Verify compatibility before specifying:
- Hot-dip galvanizing: Steel and iron only (aluminum melts at galvanizing temperatures)
- Anodizing: Aluminum, titanium, magnesium—not steel
- Powder coating: Steel, aluminum, and other conductive metals
- Electroplating: Requires conductive substrate; careful surface preparation for aluminum
Substrate incompatibility catches many projects off-guard late in the design phase—confirming material and coating pairings early prevents costly rework and schedule delays.
Factor in Performance Requirements and Total Cost of Ownership
Upfront coating cost should be weighed against expected service life, maintenance frequency, and downtime risk. Here's how common coating types stack up on a total cost basis:
- Powder coating — 15-25 years of service life vs. liquid paint's 5-15 years; higher upfront cost, but fewer recoat cycles
- Hot-dip galvanizing — 72+ years of maintenance-free protection on structural steel, eliminating recurring access, prep, and labor costs
- HVOF thermal spray / PVDF fluoropolymers — premium pricing is justified in extreme wear environments or where coating failure carries safety or operational consequences
Running a full TCO calculation often shifts the decision. A coating that costs twice as much upfront but lasts four times longer—with no scheduled maintenance—typically wins on a 20-year asset life.
Frequently Asked Questions
What are the types of metal coating?
The main categories are powder coating, hot-dip galvanizing, electroplating, liquid paint/spray coating, anodizing, and thermal spray. The right type depends on the metal substrate, environmental exposure, and performance requirements — corrosion resistance, wear resistance, or aesthetics.
What is the best metal coating?
The best coating depends entirely on the application. Galvanizing excels for structural steel in wet outdoor environments, powder coating suits fabricated parts needing color and durability, and anodizing is the go-to for aluminum in architectural or electronic use.
What is the coating on metal called?
The coating on metal is generally referred to as a metal coating or metal finish. Specific coatings have their own names based on material or process, such as galvanization, electroplating, powder coating, or anodized finish.
Does metal coating prevent rust?
Yes, most metal coatings prevent rust by creating a barrier that blocks moisture and oxygen from reaching the substrate. Hot-dip galvanizing provides additional sacrificial protection—the zinc layer corrodes preferentially to protect the underlying steel even if the coating is scratched.
What is the difference between metal coating and metal finishing?
Metal coating adds a new layer of material (protective or functional) to the surface, while metal finishing alters the surface characteristics of the existing metal through processes like polishing or brushing. Some processes like anodizing blur this line by converting the surface into a protective oxide layer.
How long does metal coating last?
Lifespan varies widely by coating type and environment. Galvanized coatings can last 20–50+ years, powder coatings typically 15–25 years, and liquid paint may need touch-up within 5–10 years depending on exposure.

