
Introduction
Manufacturing teams face a costly reality: quality failures drain budgets, delay shipments, and erode customer trust. Yet many organizations conflate "inspection" and "quality control," creating dangerous gaps in their processes.
Some operations over-rely on end-of-line checks, catching defects only after value has been added. Others run upstream controls but skip the verification checkpoints that confirm those controls are actually working.
The distinction between these two approaches directly affects defect rates, rework costs, and compliance outcomes. In precision metal fabrication—where tolerances are measured in thousandths of an inch and weld integrity determines structural safety—confusing the two has real operational consequences. This guide breaks down how quality control and inspection differ in scope, timing, and purpose, and how to use both effectively.
TL;DR
- Quality control (QC) is a systematic framework of processes that ensures products meet standards throughout production
- Inspection is a specific checkpoint activity where products are physically examined for defects at defined stages
- QC focuses on prevention and process improvement; inspection focuses on detection at a point in time
- Both are necessary: inspection without QC misses root causes; QC without inspection lacks verification data
- For custom metal fabrication, pairing systematic QC with CMM inspection tools delivers dimensional accuracy and on-spec parts
Quality Control vs. Inspection: Quick Comparison
| Attribute | Quality Control | Quality Inspection |
|---|---|---|
| Definition/Scope | Broad system governing entire production lifecycle | Specific activity at defined checkpoints |
| Timing | Ongoing throughout production | Periodic at pre-production, in-process, or pre-shipment stages |
| Primary Goal | Defect prevention and process consistency | Defect detection and conformance verification |
| Responsibility | Quality management team, process engineers | Inspectors, quality engineers |
| Tools/Methods | SOPs, process audits, training, Statistical Process Control (SPC), equipment calibration | CMM, calipers, micrometers, visual checks, weld inspection |
| Output | Documented quality management framework | Inspection report with pass/fail status and measurement data |
Inspection sits inside the quality control system — one specific tool within a broader framework, not a separate discipline running alongside it. Manufacturers with strong QC feed inspection findings back into their processes, using the data to eliminate root causes rather than simply filtering out defective parts.
What is Quality Control in Manufacturing?
Quality control in manufacturing is the system of planned activities, standards, and procedures a facility uses to ensure every product meets defined requirements. According to the American Society for Quality (ASQ), QC spans materials, processes, personnel, and equipment — covering the entire production environment.
The Three Pillars of QC
Effective quality control rests on three foundational pillars:
- Setting quality standards aligned with customer specifications and regulatory requirements
- Monitoring and controlling production processes to stay within acceptable limits using techniques like Statistical Process Control
- Identifying and correcting deviations before they produce nonconforming output

Business Impact of Strong QC Systems
Research shows that the Cost of Poor Quality (COPQ) consumes 15% to 20% of annual sales revenue for average manufacturers, with extreme cases reaching up to 40% of total operations costs. By contrast, best-in-class organizations with effective quality management programs spend 38% less on their total cost of quality compared to peers.
The operational benefits show up directly in business outcomes:
- Lower rework and scrap costs when defects are caught early, before they compound into larger failures
- Improved throughput as process consistency reduces the variability that creates production bottlenecks
- Reduced exposure to regulatory penalties and product recalls through documented compliance records
QC vs. QA: Understanding the Layers
Quality control is the execution layer—what workers do on the shop floor to verify products meet requirements. Quality assurance (QA) is the management layer—the policies, audits, and system reviews that confirm the QC system is functioning as intended. Inspection sits under QC as one of several operational techniques used to verify conformance.
Use Cases of Quality Control
QC operates throughout the production lifecycle:
- Raw material qualification – Verifying material certifications (MTRs) and incoming material properties before production begins
- Process parameter monitoring – Using Statistical Process Control (SPC) to track variables like cutting speeds, temperatures, and pressures in real time
- Equipment calibration schedules – Maintaining measurement system accuracy through regular calibration and gage R&R studies
- Operator training protocols – Ensuring personnel understand specifications, measurement techniques, and quality standards
- Corrective action systems (CAPA) – Documenting nonconformances and implementing root cause corrections
Industries where rigorous QC systems are essential include:
- Aerospace (AS9100) – Requiring strict traceability, counterfeit parts prevention, and First Article Inspection compliance
- Medical devices (ISO 13485) – Emphasizing risk management and process validation for patient safety
- Automotive (IATF 16949) – Mandating advanced core tools like APQP, PPAP, FMEA, and SPC
- Custom metal fabrication – Governing dimensional tolerances, weld integrity, and surface finish requirements
In high-stakes manufacturing, a documented QC system is not optional. It is a prerequisite for supplier qualification.
What is Quality Inspection in Manufacturing?
Quality inspection is the act of measuring, examining, or testing a product, component, or material against defined criteria at a specific point in the production process to determine conformance or non-conformance. Unlike quality control, which is a continuous system, inspection is an event—a checkpoint that captures a conformance snapshot at one point in time.
Three Primary Inspection Stages
Pre-Production Inspection (PPI):Conducted before production begins, PPI verifies raw materials, component quality, and setup readiness. This stage catches issues at the source—confirming material certifications match specifications, checking tooling setup, and validating that the production environment is ready to produce conforming parts.
During Production Inspection (DUPRO/In-Process):Performed when 20% to 80% of production is complete, DUPRO monitors output mid-run to catch process drift or tool wear early. Research demonstrates that inline inspection prevents defect propagation and allows for immediate corrective actions before entire batches are affected.
Pre-Shipment Inspection (PSI):The final conformance check before product leaves the facility, PSI verifies that finished goods meet specifications, packaging is correct, and documentation is complete. This acts as the last safeguard against shipping nonconforming parts to customers.
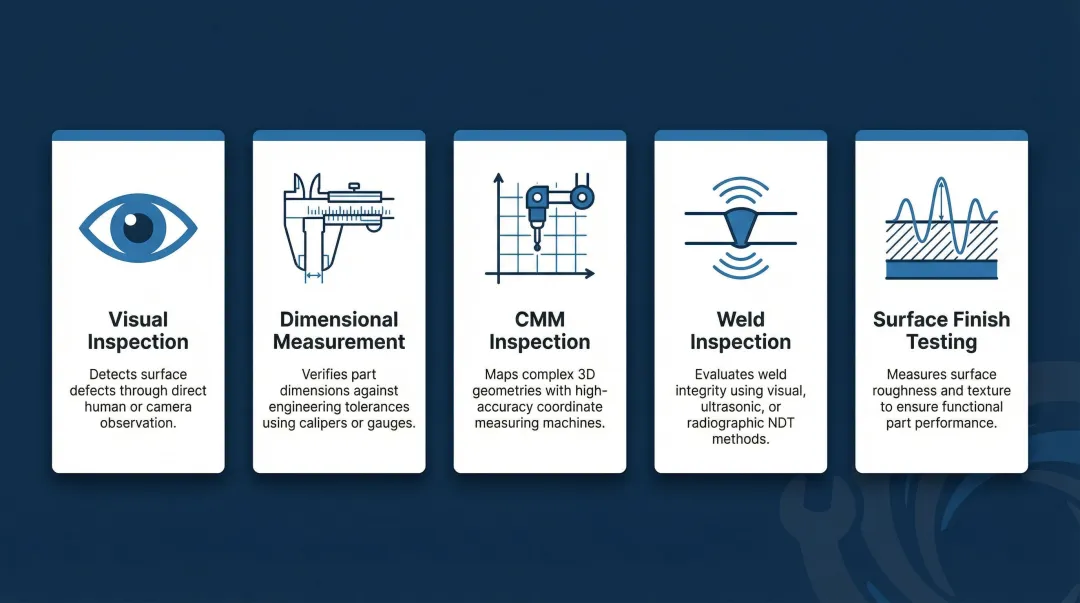
Common Inspection Methods in Manufacturing
- Visual inspection – Identifies surface defects, weld quality issues, and finish anomalies without specialized equipment
- Dimensional measurement – Calipers, micrometers, and height gauges verify basic features against engineering tolerances
- CMM inspection – Coordinate Measuring Machines validate complex geometries and GD&T characteristics with sub-micron accuracy
- Weld inspection – Combines visual examination with NDT methods like ultrasonic or dye penetrant testing to detect subsurface flaws
- Surface finish testing – Quantifies roughness, coating thickness, and adhesion properties after finishing operations
Method selection depends on part complexity, tolerance requirements, and applicable industry standards. IMTS, for instance, operates the Keyence XM-5000 Portable CMM, Starrett HGC 1820-16 stationary CMM, and Keyence IM-8000 imaging measurement system to generate full inspection reports with documented conformance data for every job.
The Limits of Inspection as a Standalone Activity
Inspection finds defects after they've already been made—it does not prevent them. As quality pioneer W. Edwards Deming stated, "Inspection is too late. The quality, good or bad, is already in the product". Relying solely on end-of-line inspection is reactive and expensive because it does nothing to correct the root cause of variation.
There's another hard limit: 100% inspection does not guarantee 100% defect detection due to measurement system uncertainty, gauge repeatability limitations, and human factors like fatigue. Inspection only delivers real value when backed by a QC system that uses inspection data to drive process improvements.
Use Cases of Quality Inspection
Inspection adds the most value at these stages in a fabrication workflow:
- First-article verification – Confirming that initial production parts meet all specifications before committing to a full production run
- Critical weld joint checks – Validating structural welds on load-bearing components using visual and NDT methods
- CNC-machined part verification – Confirming dimensional accuracy of complex features against engineering drawings using CMM inspection
- Surface finish validation – Measuring coating thickness and finish quality after powder coating or media blasting
Example use case: A shop producing precision brackets for industrial equipment uses in-process CMM checks after CNC milling to verify critical hole locations and profile tolerances before parts move to welding and finishing. Catching deviations at this stage—when rework involves only re-machining—prevents the far more expensive scenario of discovering dimensional problems after welding and coating are complete.
Key Differences: How Quality Control and Inspection Work Together
The most common source of confusion: manufacturers often treat inspection as synonymous with quality control, creating a reactive-only system. The real relationship is architectural—QC is the framework; inspection is one of its diagnostic tools.
Four Key Practical Differences
1. ScopeQC covers the entire production system—from material receiving through final shipment. Inspection covers a defined product sample at a specific point in time.
2. TimingQC is continuous, operating through process monitoring, training, and maintenance schedules. Inspection is periodic, occurring at pre-defined checkpoints.
3. OutcomeQC reduces defect probability by controlling process inputs and variables. Inspection reveals current defect presence by measuring outputs.
4. Cost ImpactStrong QC lowers the total inspection burden needed because processes become more predictable and capable. The 1-10-100 Rule demonstrates this principle—every $1 spent preventing a defect saves $10 on in-process correction and $100 in post-shipment failure costs.

Situational Emphasis: When to Lean on Each
When to emphasize inspection:
- Early production runs with new tooling or materials
- Customer specifications requiring documented conformance reports
- Tight-tolerance applications in critical industries (aerospace, medical)
- First Article Inspection (FAI) requirements per AS9102 standards
When to prioritize QC investment:
- Recurring defect patterns indicating process problems, not detection problems
- Scaling up production volume where prevention becomes more cost-effective than sorting
- Pursuing certifications like ISO 9001, AWS, or IATF 16949
- Building supplier qualification capabilities for regulated industries
A Well-Integrated System in Practice
Knowing when to lean on each tool is only half the equation. The real gains come from connecting them deliberately.
Inspection checkpoints are embedded at strategic production stages, teams document and track results over time, and nonconformances trigger root cause analysis that feeds back into process controls. Each defect caught becomes an input for improving the process that allowed it.
At IMTS, this shows up concretely: CMM inspection data from both portable and stationary equipment informs process adjustments on the shop floor, not just end-of-run compliance reporting. AWS-certified welding procedures and integrated inspection at each fabrication stage reflect how QC and inspection reinforce each other when the system is built intentionally from the start.
Conclusion
Quality control and inspection are not competing approaches—they are complementary layers of a sound manufacturing quality system. Run inspection alone and you're catching problems after material, time, and labor have already been consumed. Run QC without inspection and you're trusting that processes are working correctly without ever verifying it. Each approach is incomplete without the other.
Manufacturers who understand this relationship invest in both, calibrating the depth of inspection to the risk level of each production stage. For custom fabricators and their customers, a documented QC system backed by precise inspection capability—CMM reports, dimensional checks at critical stages—translates directly into fewer rejected parts, less rework, and shorter lead times. Those gains compound over time: tighter supplier relationships, more predictable scheduling, and a track record that makes future quoting easier for everyone involved.
Frequently Asked Questions
Is quality inspection the same as quality control?
No. Inspection is a specific activity performed at defined checkpoints to detect defects and verify conformance. Quality control is the broader system of processes, standards, and controls that governs the entire production lifecycle. Inspection is one component of QC, not a synonym for it.
What are the main types of quality inspections in manufacturing?
Three primary types cover the production lifecycle:
- Pre-Production Inspection (PPI) — verifies materials and setup before production begins
- During Production Inspection (DUPRO) — monitors output mid-run to catch drift early
- Pre-Shipment Inspection (PSI) — confirms final conformance before delivery
What is the difference between quality control and quality assurance?
QC is the operational layer—the hands-on activities including inspection that verify products meet requirements. QA is the management layer that plans, audits, and reviews the overall quality system to ensure it is functioning as intended and continuously improving.
When should a manufacturer prioritize inspection over broader QC improvements?
Heavier inspection is appropriate during new product launches, first article qualification, tight-tolerance applications, or when customers require documented conformance data. However, recurring defects signal a process issue — one QC investment must address at the source, not just catch at inspection.
What tools are used for quality inspection in metal fabrication?
Common tools include:
- CMM (coordinate measuring machines) — dimensional accuracy and GD&T verification
- Calipers and micrometers — basic dimensional measurements
- Weld inspection methods — visual examination and non-destructive testing
- Surface finish gauges — coating thickness and roughness verification
Can a small manufacturer implement both quality control and inspection effectively?
Yes. Scalable QC does not require enterprise-level infrastructure. It starts with documented standards, defined inspection checkpoints, and a system for logging and acting on nonconformances. These elements can be built incrementally as production volume grows, with investment prioritized based on risk and customer requirements.


